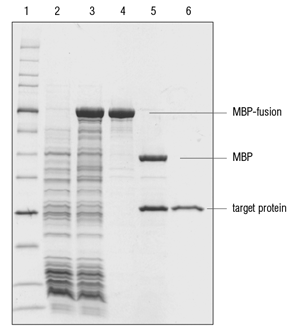
Recombinant Mbp Mcp Conjugated Amylose Resin (New England Biolabs) | Bioz | Ratings For Life-Science Research

Purification strategy for the ubiquitylated proteins. The main steps... | Download Scientific Diagram

Presence and removal of a contaminating NADH oxidation activity in recombinant maltose-binding protein fusion proteins expressed in Escherichia coli | BioTechniques

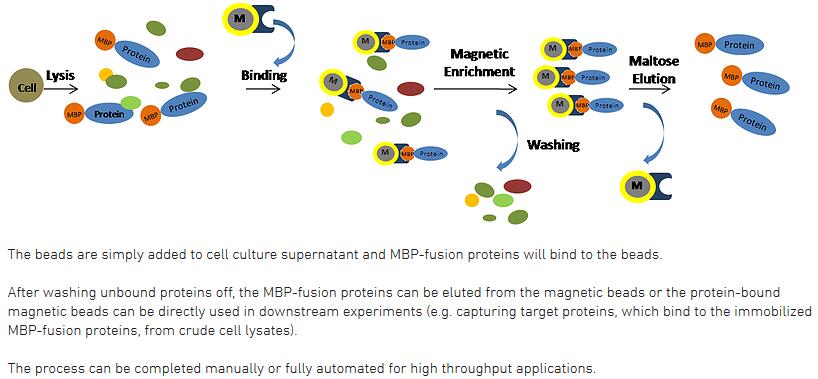
Protein and antibody purification followed by immunoprecipitation of MYB and GATA zinc finger-type maize proteins with magnetic beads - ScienceDirect

An efficient method for FITC labelling of proteins using tandem affinity purification. - Abstract - Europe PMC
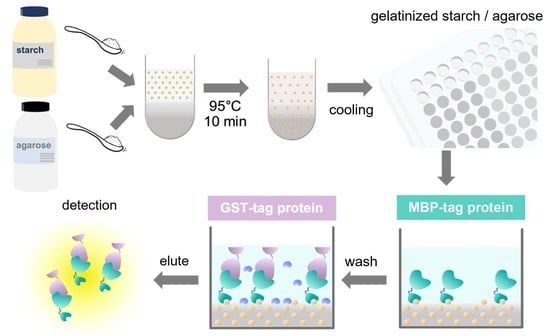
MPs | Free Full-Text | A Cost-Effective Immobilization Method for MBP Fusion Proteins on Microtiter Plates Using a Gelatinized Starch–Agarose Mixture and Its Application for Convenient Protein–Protein Interaction Analysis

Construction of redesigned pMAL expression vector for easy and fast purification of active native antimicrobial peptides | bioRxiv

Presence and removal of a contaminating NADH oxidation activity in recombinant maltose-binding protein fusion proteins expressed in Escherichia coli | BioTechniques

Purification of hetero-oligomeric protein variants using a modified tandem affinity purification approach - ScienceDirect
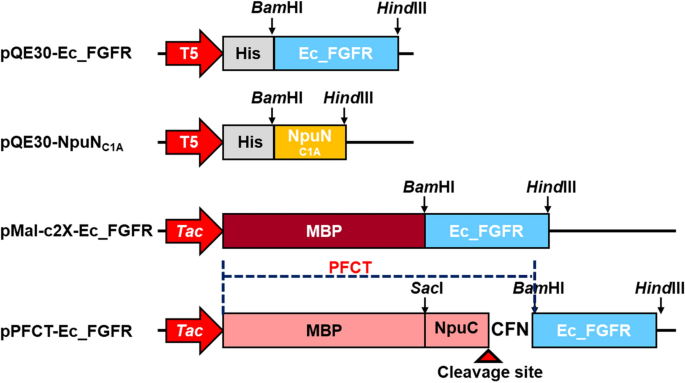
A designed fusion tag for soluble expression and selective separation of extracellular domains of fibroblast growth factor receptors | Scientific Reports

One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text
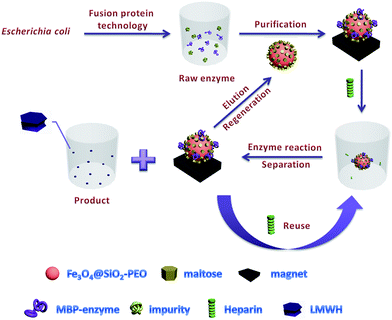
Magnetic nanoparticles for the affinity adsorption of maltose binding protein (MBP) fusion enzymes - Journal of Materials Chemistry (RSC Publishing) DOI:10.1039/C2JM16778F